
Introduction
HealthTech companies in 2026 operate under a compliance landscape that has grown substantially more complex. HIPAA and HITECH set the baseline, but new CMS mandates, state-level privacy laws, and AI governance rules now layer on top. Manual processes—spreadsheets, email chains, quarterly audits—no longer keep pace with this regulatory complexity.
The average US healthcare data breach now costs $7.42 million, with healthcare remaining the costliest industry for breaches for 14 consecutive years. HIPAA civil penalties reach up to $2,134,831 per violation category annually under Tier 4 (willful neglect, uncorrected). For HealthTech companies racing to scale, regulatory fines and operational disruption can stall growth entirely.
That's the context for this guide. It covers what healthcare regulatory compliance technology (RegTech) actually is, which frameworks apply to HealthTech companies right now, the core compliance tech categories available in 2026, and what it takes to build a compliance program that keeps your business moving without exposing your patients.
TLDR
- RegTech platforms automate adherence to HIPAA, HITECH, state privacy laws, and CMS mandates through centralized documentation and real-time monitoring
- Core tools span policy management, automated risk assessment, compliance dashboards, LMS platforms, and vendor risk systems
- Major 2026 trends: AI-powered predictive monitoring, cloud-based platforms, and enhanced cybersecurity tooling
- Tools only work when skilled compliance professionals configure and act on them — and that talent is genuinely hard to find in HealthTech
What Is Healthcare Regulatory Compliance Technology — and Why HealthTech Companies Can't Ignore It in 2026
Healthcare regulatory compliance technology (RegTech) refers to cloud-based, automated, or AI-assisted platforms that help healthcare and HealthTech organizations track adherence to legal, operational, and safety standards. These systems replace manual processes — spreadsheets, email chains, quarterly checklists — with centralized, auditable platforms that provide live compliance visibility across the organization.
What Makes HealthTech Compliance Different
Unlike established hospitals or health systems with legacy compliance infrastructure, HealthTech startups face distinct challenges:
- Multi-state licensing complexity from day one — telehealth platforms must comply with medical board requirements across every state where they serve patients
- Dual compliance obligations — companies must satisfy both clinical requirements (HIPAA, CMS) and commercial obligations (state privacy laws, consumer protection rules)
- Limited internal expertise — early-stage companies rarely have dedicated compliance officers, yet they face the same regulatory scrutiny as established health systems
What Compliance Technology Manages
RegTech platforms address a broad mandate:
- Policy documentation and version control
- Regulatory training and certification tracking
- Incident reporting and breach notification
- Risk assessments and vulnerability monitoring
- Vendor management and Business Associate Agreement (BAA) tracking
- Audit preparation and evidence collection
The Cost-of-Inaction Case
Healthcare organizations devote enormous resources to compliance. Clinicians now spend nearly 28 hours per week on administrative duties, with 82% reporting burnout symptoms. Administrative costs account for more than 40% of total hospital expenses.
Non-compliance carries multi-million-dollar consequences. OCR collected $7,860,566 across 18 enforcement actions in 2025, with individual settlements reaching $2.175 million. The average breach takes 279 days to contain and costs $7.42 million — numbers that dwarf the cost of any compliance platform. The sections below break down what to look for when selecting one.
Key Regulatory Frameworks Every HealthTech Company Must Know in 2026
HIPAA and HITECH
HIPAA's Privacy Rule, Security Rule, and Breach Notification Rule establish how protected health information (PHI) must be handled. HITECH strengthens enforcement through tiered civil and criminal penalties and mandates EHR adoption.
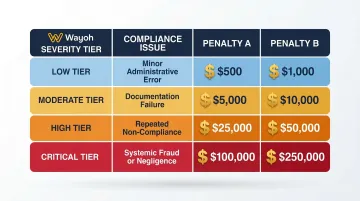
Penalty structure (effective August 2024):
| Tier | Culpability | Max per Violation | Annual Cap |
|---|---|---|---|
| 1 | Lack of Knowledge | $71,162 | $2,134,831 |
| 2 | Reasonable Cause | $71,162 | $2,134,831 |
| 3 | Willful Neglect (corrected) | $71,162 | $2,134,831 |
| 4 | Willful Neglect (uncorrected) | $2,134,831 | $2,134,831 |
HealthTech companies—telehealth platforms, digital health apps, remote care tools—fall squarely under HIPAA as either covered entities or business associates, carrying the full weight of its obligations.
The 21st Century Cures Act and Information Blocking
This law prohibits practices that interfere with the access, exchange, or use of electronic health information. HealthTech companies building EHR-connected platforms must ensure interoperability through compliant APIs.
Key details:
- Enforcement began September 1, 2023 for health IT developers
- Penalties reach $1 million per violation for HIT developers
- Enforcement extended to healthcare providers July 1, 2024
- Eight codified exceptions exist (Preventing Harm, Privacy, Security, Infeasibility, Health IT Performance, Content and Manner, Fees, Licensing)
CMS Mandates and Network Adequacy Requirements
CMS has introduced stricter rules around provider directory accuracy, appointment wait times, and network adequacy. For HealthTech platforms operating within Medicare Advantage or Medicaid networks, this includes:
- CMS-4208-F2: Finalized rule establishing provider directory requirements beginning October 1, 2026 for Plan Year 2027
- REAL Health Providers Act (S.3750): Proposed legislation to establish provider directory accountability
State Privacy Laws — Beyond HIPAA
Federal law sets the floor, but several states have enacted health privacy rules that reach further — and carry independent enforcement teeth:
Washington's My Health My Data Act (effective March 31, 2024):
- Applies to any entity conducting business in Washington or targeting Washington consumers
- No revenue thresholds; nonprofits not exempt
- Broad private right of action
- Opt-in consent for collection; separate consent for sharing
- Geofencing prohibition around healthcare facilities
Texas HB 300 (Chapter 181):
- Covers any person or entity that assembles, collects, analyzes, uses, or transmits PHI
- Penalties up to $250,000 per intentional violation
- Annual cap of $1.5 million for patterns of violation
- Required training within 90 days of hire; repeated every 2 years
California Privacy Rights Act (CPRA):
- Classifies health data as sensitive personal information
- HIPAA-governed PHI exempt
- Enforced by California Privacy Protection Agency
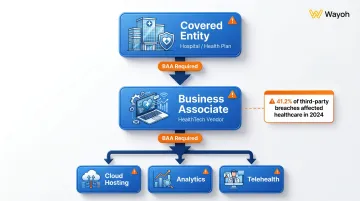
Who Actually Needs to Comply — The Liability Chain
HIPAA extends to covered entities (providers, health plans) and all business associates—including subcontractors. HealthTech vendors, cloud hosting partners, analytics firms, and telehealth platforms are all on the hook. That liability runs down the entire vendor chain — HealthTech companies are responsible for auditing every third party they contract with.
Healthcare accounted for 41.2% of all third-party breaches tracked in 2024. The Change Healthcare ransomware attack alone compromised data for approximately 190 million individuals — a reminder that compliance gaps don't stay theoretical for long.
Core Categories of Healthcare Compliance Technology
Policy and Document Management Platforms
These tools serve as a central, version-controlled repository for compliance policies, procedures, and regulatory documents. Features include:
- Automated distribution to staff
- Employee attestation tracking
- Update alerts when regulations change
- Audit trails showing who accessed what and when
For HealthTech companies updating policies frequently as regulations evolve, centralized policy management prevents the common failure mode: outdated policies circulating via email while current versions sit on someone's hard drive.
Automated Risk Assessment and Monitoring Tools
Risk assessment software identifies, scores, and tracks compliance vulnerabilities across an organization: both technical gaps (unpatched systems, insecure data storage) and process gaps (missing vendor contracts, expired certifications).
Continuous monitoring is the 2026 standard. Point-in-time audits conducted quarterly or annually no longer suffice as HealthTech companies expand vendor networks. Platforms like Clearwater Security's IRM|Pro provide ongoing risk scoring that surfaces issues before they become enforcement actions.
Real-time Compliance Dashboards and Audit Readiness
Once risk gaps are tracked, they need somewhere to live — and dashboards deliver that visibility. Compliance dashboards consolidate key performance indicators into a single view for compliance officers and executives:
- Policy acknowledgment rates
- Training completion percentages
- Audit scores and findings
- Incident resolution timelines
- Vendor risk scores
Critical distinction: Compliance and audit-readiness are not the same thing. Organizations can meet daily requirements but still fail regulatory surveys if documentation is scattered, evidence is incomplete, or staff cannot answer auditor questions. Dashboards keep documentation organized by control area and accessible within minutes — not scrambled together the week before a survey.
Learning Management Systems (LMS) for Compliance Training
LMS platforms automate training assignment, track completion and certification, and offer role-specific course libraries. HealthStream and Relias provide pre-built HIPAA, OSHA, and CMS content designed by regulatory experts.
Ongoing staff training is both a regulatory requirement under HIPAA and a practical necessity. Many violations result from employees being unaware they're breaking compliance rules—accessing records without authorization, emailing PHI insecurely, or discussing patient information in public areas.
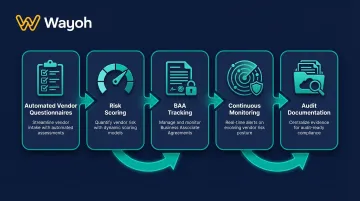
Third-Party Risk Management (TPRM) Tools
TPRM platforms help HealthTech companies evaluate, monitor, and document the compliance posture of business associates and vendors. Given that 41.2% of third-party breaches affected healthcare in 2024, vendor oversight has become one of the highest-leverage compliance investments a HealthTech company can make.
Key features include:
- Automated vendor questionnaires
- Risk scoring based on security controls
- Business Associate Agreement (BAA) tracking
- Continuous monitoring for vendor breaches
- Documentation for regulatory audits
2026 Compliance Technology Trends Reshaping HealthTech
AI and Predictive Compliance Monitoring
AI-powered compliance tools now flag potential violations before they occur — not after the damage is done. Modern platforms do this by:
- Analyzing historical patterns to identify recurring risk areas
- Detecting anomalies in data access (such as bulk EHR exports at unusual hours)
- Forecasting audit exposure before regulators come knocking
- Automating internal audits to cut manual review time and labor costs
91% of healthcare providers expressed optimism about generative AI reducing administrative burden, according to a 2024 Google Cloud/Harris Poll survey.
Blockchain for Credentialing and Provider Verification
Blockchain technology creates tamper-proof records of provider credentials, licenses, and sanctions. This addresses a persistent challenge for HealthTech platforms that onboard clinical providers at scale and need to maintain continuous credentialing compliance.
Adoption is still early, but blockchain-based credentialing systems make fraudulent record alterations far harder to pull off — and give payers and regulators a path to instant verification.
Cloud-Based Compliance Platforms and Mobile-First Access
Cloud-based solutions enable compliance teams across geographies to collaborate in real time, access centralized documentation, and maintain secure data storage. The rise of telehealth and remote care platforms has accelerated demand for mobile-friendly compliance interfaces.
As a result, clinical and administrative staff can now complete training, attestations, and reporting from anywhere — a practical necessity for distributed teams spanning multiple states and time zones.
Common Implementation Challenges — and How HealthTech Companies Can Overcome Them
Integration with Legacy Systems and Existing Tech Stacks
Many HealthTech companies—especially those that have grown through acquisition or rapid scaling—face challenges connecting new compliance platforms with existing EHRs, HR systems, and data infrastructure.
Prioritize platforms with strong API capabilities and pre-built integrations. Roll out one solution at a time rather than overhauling everything at once.
Cost, Resource Allocation, and Early-Stage Constraints
Early-stage HealthTech companies often lack dedicated compliance budgets and must balance technology investment against staffing costs.
The cost of non-compliance almost always outweighs the cost of a compliance platform. At $7.42 million per breach versus typical RegTech costs of $10,000–$50,000 annually, the math is straightforward. Scalable SaaS tools let startups right-size their investment as they grow — and under-resourced teams that cut compliance corners often face a harder problem: incomplete documentation.
Inadequate Documentation and Evidence Tracking
Missing documentation is the most common compliance program failure. Regulatory auditors treat missing evidence as missing compliance — full stop.
OCR stated in a July 2025 settlement: "Common deficiencies include lacking a risk analysis entirely or failing to update existing risk analyses when implementing new technologies."
Centralized compliance platforms address this directly. They automatically timestamp and attribute every compliance activity, generating regulator-ready audit trails without manual spreadsheet maintenance.
Building Your Compliance Foundation: Technology and the Right Team
Compliance technology is only as effective as the people managing it. Platforms require skilled professionals who understand how to configure risk frameworks, interpret dashboard signals, manage vendor relationships, and translate regulatory updates into operational policy changes.
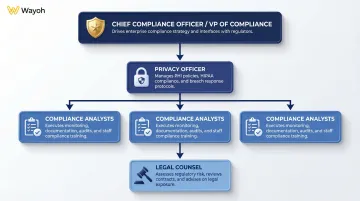
The Compliance Roles HealthTech Companies Need
As HealthTech companies scale, they typically need:
- Chief Compliance Officer or VP of Compliance — sets strategy, interfaces with regulators, owns the compliance program
- Privacy Officer — HIPAA and state law expertise, manages PHI policies and breach response
- Compliance analysts — day-to-day monitoring, documentation, training coordination
- Legal counsel — HealthTech-specific regulatory risk, contract review, enforcement response
The timing and sequencing of these hires depends on the company's stage, regulatory exposure, and whether they operate in clinical settings.
The Talent Challenge
Finding compliance professionals with deep HealthTech experience—people who understand HIPAA, state licensing, CMS requirements, and the clinical-commercial intersection—is a genuine challenge. The BLS projects only 3% growth for compliance officers from 2024-2034, classified as "slower than average," despite expanding regulatory complexity.
That supply-demand gap makes search quality critical. Wayoh has spent 10+ years placing compliance professionals in HealthTech, FinTech, and financial services — with direct knowledge of HIPAA, state licensing, CMS requirements, and the clinical-commercial distinctions that define these roles. Every search draws on personal networks built over years, not keyword filters applied to a database.
Frequently Asked Questions
What is regulatory compliance in healthcare technology?
Healthcare regulatory compliance technology (RegTech) refers to software platforms and automated tools that help organizations adhere to laws like HIPAA, HITECH, and CMS mandates by centralizing documentation, automating risk assessments, and generating audit-ready records.
What is an example of RegTech?
Examples include automated HIPAA compliance dashboards that monitor PHI access in real time, AI-powered policy management tools that flag outdated procedures, and vendor risk management platforms that track Business Associate Agreement compliance across a HealthTech company's third-party network.
What regulations apply specifically to HealthTech startups in 2026?
HealthTech startups typically fall under HIPAA, HITECH, and the 21st Century Cures Act's information blocking rules. Companies operating in Medicare/Medicaid networks also face CMS mandates, and state privacy laws — including CPRA and Washington's My Health My Data Act — apply based on where patients are served.
What is the difference between HIPAA and HITECH?
HIPAA sets foundational standards for protecting PHI through the Privacy Rule, Security Rule, and Breach Notification Rule. HITECH strengthens HIPAA's enforcement by introducing tiered civil and criminal penalties, expanding breach notification requirements, and incentivizing electronic health record adoption.
How does a HealthTech company know if it needs a dedicated compliance officer?
Any HealthTech company that handles PHI, operates as a business associate under HIPAA, or faces CMS or state licensing requirements should appoint a dedicated compliance officer. The need becomes critical at Series A and beyond, once regulatory complexity and vendor relationships make informal compliance management unsustainable.
What compliance roles should a HealthTech company hire first?
Most HealthTech companies should prioritize a Privacy Officer or Chief Compliance Officer as their first dedicated hire, followed by a compliance analyst to handle documentation and training. Clinical operations typically require a credentialing specialist and HIPAA-experienced legal counsel as well.


